The immune response is a double-edged sword; it is absolutely required for host defense, but unregulated, cause inflammatory disease. Diverse and potent mechanisms have evolved to recognize and counter invading microorganisms. A number of critical questions regarding the mechanisms that appropriately tailor these pathways remain unanswered. In order to answer these questions is necessary to acquire complete knowledge of all the components of the cellular machinery and an understanding of how the architectural arrangement of these components leads to the appropriate coordination of host defense. Furthermore, while significant effort has been directed at understanding how immune responses are initiated, much less is known about how these robust and self-perpetuating mechanisms are constrained. This proposal seeks to uncover novel regulatory mechanisms that control critical aspects of the innate and adaptive response to NIAID priority pathogens.
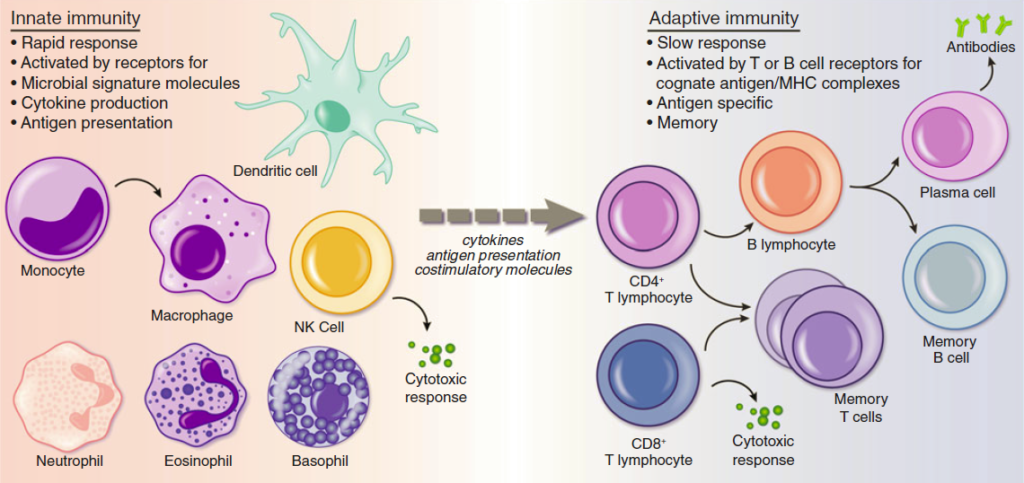
Figure from Review by Beutler and Goodnow. Mamm Genome (2011)
The Innate Immune Response
The innate immune system is the first line of defense against infectious agents. In order for innate immune cells to discriminate the large number of potential pathogens from self, they have evolved a variety of pattern recognition receptors, including Toll-like receptors (TLRs), RIG-I-like receptors (RLRs), Nod-like receptors (NLRs), and C-type lectin-like receptors (CLRs) that recognize conserved molecular motifs on pathogens. Although many components of the signaling pathways activated by these receptors have been identified, numerous critical questions remain including how signals propagate following receptor engagement, how these signaling interactions drive transcriptional, post-transcriptional, and post-translational changes, and how innate immune responses are appropriately modulated and ultimately shut down. These responses in macrophages and dendritic cells are crucial to host defense not only for immediate control of invading microorganisms but also for subsequent instruction of the adaptive immune system.
The Adaptive Immune Response
Effective adaptive immune responses hinge upon the successful execution of separate processes for short- and long-term increases in the activity of antigen-specific lymphocytes. Swift multiplication of effector CD8 T cells and plasma cells is critical to control an immediate infection, but this must not be at the expense of establishing a pool of antigen-specific memory T and B cells that maintain immunity for years and mount an accelerated response upon re-exposure. Long-term immunity depends upon antibody affinity maturation in germinal centers, and this process is intimately linked to the formation of long-lived memory B cells and plasma cells. A central unanswered question in the field concerns how B cells make decisions between differentiation into short-lived plasma cells, germinal center B cells, memory B cells, and long-lived plasma cells. Equally, high interest currently focuses on how CD8 T cells decide between differentiation into short-lived cytotoxic T cells and long-lived memory CD8 cells.
The Scope of Our Investigations
Beyond the context of infection, these studies will lead to a deeper understanding of the mechanisms that underlie the balance between host defense and immunologic disease. Inflammation is directly responsible for many human diseases including lupus and rheumatoid arthritis. Furthermore, certain diseases once considered non-inflammatory such as atherosclerosis and Alzheimer’s disease are dependent on proteins and pathways involved in inflammatory responses. Currently, most anti-inflammatory therapies act broadly and consequently suppress immune responses rendering the patient susceptible to infection. An ideal anti-inflammatory drug would therefore be highly specific in its action. By defining regulatory nodes in the molecular networks that control immune responses we could ultimately expect to treat both sterile and infectious inflammation far more effectively, and far more specifically. The long-term goal of this program is to provide new approaches for the development of improved vaccines or therapeutics directed against human disease caused by emerging/re-emerging pathogens.